Hydrocarbons
What is a Hydrocarbon?
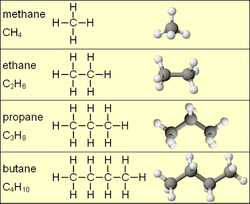
A hydrocarbon is an organic molecule compound that consist entirely of carbon and hydrogen. Most hydrocarbons are found in crude oil where decomposing materials release hydrogen and carbon. There are three main types of hydrocarbons: Alkene, Alkane, and Alkyne. Alkene hydrocarbons have a single bond, Alkane have a double bond, and Alkyne have a triple bond. The amount of carbons in a hydrocarbon determines the name of the compound. An alcoholic hydrocarbon consists of the regular structure with an oxygen and hydrogen atom bonded to the end. A cyclo hydrocarbon is a nonlinear hydrocarbon that bonds to form a cirlce. The ten main hydrocarbons are methane, ethane, propane, butane, pentane, hexane, heptane, octane, nonane, and decane: With methane having one carbon and decane have 10 carbons. Hydrocarbons are a combustible fuel source. In their solid form they are asphalt. Some uses of hydrocarbons are: Butane (C4H10) for lighters, propane (C3H8) for barbecuing, and methane (CH4) for cooking. As you can see, hydrocarbons are very important in our everyday lives.
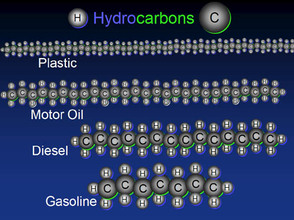
These are a few of some of the more complex hydrocarbons and their uses.
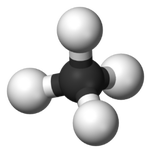
This is a simple methane hydrocarbon (CH4).